Last Updated: March 2025 (Government and Biotechnology)
Table of Contents
Government and Biotechnology
This article deals with ‘Government and Biotechnology – UPSC.’ This is part of our series on ‘Science and Technology’ which is an important pillar of the GS-3 syllabus. For more articles, you can click here.
Department of Biotechnology
Department of Biotechnology is the nodal ministry responsible for issues related to biotechnology in India.
Timeline
| 1982 | National Biotechnology Board was set up by the government. |
| 1986 | It was replaced by the Department of Biotechnology under the Ministry of Science and Technology. |
Aim
- Promote the development of modern biology and biotechnology in the country.
- Promoting international cooperation in the field of biotechnology.
- Promoting manufacturing activities.
Department of Biotechnology has also set up BIRAC (Biotechnology Industry Research Assistance Council) Agency to strengthen and empower emerging biotechnology enterprises to undertake strategic research and innovation.
Present Status of Biotechnology Sector in India

- Biotech Sector is one of the sunrise sectors in India.
- Within the Indian biotechnology sector, the major share is that of the biopharma sector accounting for a massive 55% of revenues while bioagri is the distant second with a 22% share.
- Indian Biotech Sector holds about a 3% share of the global biotech industry.
- Indian Biotech Industry is valued at $ 11 Billion and ranked 12th globally.
- India has emerged as a leading destination for clinical trials, contract research and manufacturing activities.
Some Indian Companies
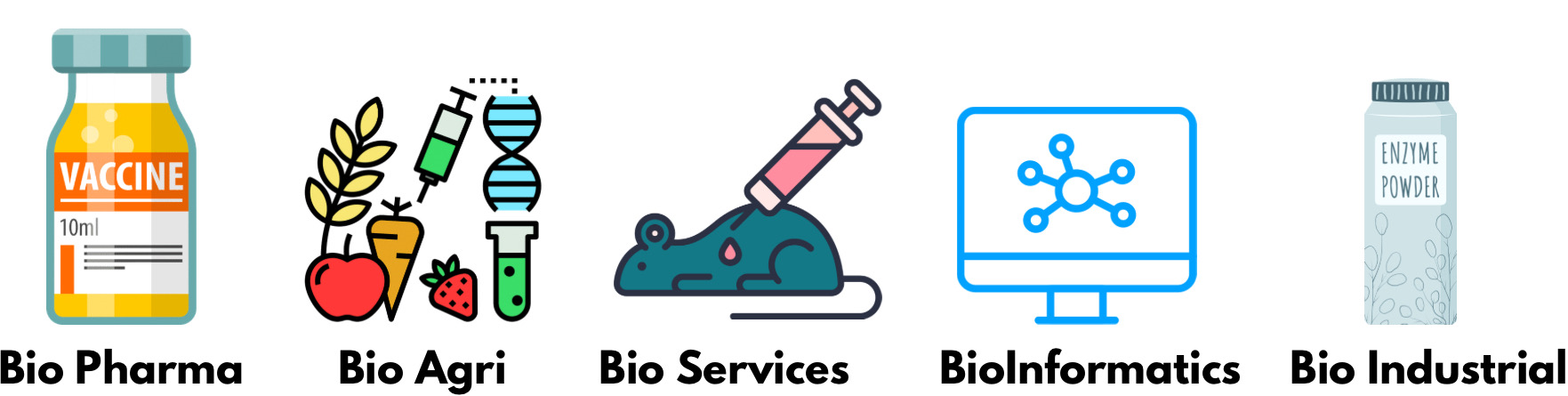
The biotech sector comprises five segments – Biopharma, Bio-services, Bio-Agri, Bio-industrial, and Bioinformatics.
1. Bio-Pharma
- This industry comprises of production of vaccines, bio-similars, regenerative medicine etc.
- Biopharma has a major share in the Indian biotechnology sector, comprising 55% of revenue.
- It has to be noted that India is the supplier of 60% of global vaccines and 22% of global generic drugs.
Example of Indian companies in this sector includes
| Serum Institute of India | Largest vaccine producer in the world (including producer of Covishield). |
| Panacea | One of the largest biopharma companies and producer of Pentavalent Vaccine. |
| Haffkine | Haffkine Pharmaceutical (Mumbai) is famous for manufacturing oral polio vaccines and anti-snake venom. |
2. Bio-Agri
Bio-Agri companies are associated with GM seeds, bio-fertilizers, bio-pesticides etc.
Prominent companies related to this sector include
| Mahyco | – MAHYCO = Maharashtra Hybrid Seeds Company – The company was founded in 1963. – The company has collaborated with various academic institutions and industries for development in the agri-business. Its well-known collaboration is with multinational Monsanto for using Bt Technology in cotton since 1998. |
| Other | – Andhra Pradesh State Seeds Development Corporation Limited. – JK Agri Genetics Limited – Kalash Seeds Limited – Kaveri Seeds Company Limited – National Seeds Corporation Limited |
3. Bio-Services
It involves industry related to contract manufacturing and clinical research.
Leading Indian companies involved in this include
- Advinus: Tata company engaged in drug discovery and end to end bio services.
- GVK Bio: Involved in drug discovery and clinical trials.
4. Bioinformatics
Bioinformatics is the integration of biotechnology and Information Technology (IT). Indian companies in this field include
- Tata Elxsi
- iLABS (Hyderabad)
- Satyam Computer Services
- Navya Biologicals
5. Bio-Industrial
The bio-Industrial sector is mainly involved with the production of enzymes and energy generation using biomass and biofuel.
Prominent companies involved in this include
- Novozyme, Rossari Biotech and Zyrtec: Companies involved in the development and production of industrial enzymes.
- Hindustan Bioenergy, IKF Green Fuels and Indian Oil Creda Biofuels: Companies engaged in the production of biofuels.
Initiatives to develop Biotech Sector in India
1. National Biotech Development Strategy
- Aim: Develop India into Biotech Manufacturing Hub.
- Primary Focus Areas (4): Food, Health, Clean Energy & Education
- Target: Develop a $100 billion biotechnology industry in India by 2025.
- Using startups and incubation centres, this will be achieved by converting scientific studies and technology into viable commercial products.
2. National Biopharma Mission
- Under the Mission, Industry and Academia will collaborate for the development of biopharmaceuticals.
- It is assisted by World Bank to create an ecosystem to promote entrepreneurship in the biopharma sector.
- The main focus of the mission includes:-
- Development of new vaccines and therapeutics.
- Bring centres of excellence in the educational sector together to strengthen current bio clusters.
- Attract FDI in the Biopharma sector.
- Deliver atleast 6 to 10 new products in biopharma in next five years.
3. The North East States specific schemes
North East is the primary focus in Bio-Pharma sector, and there are special programs for North East like
- Phyto-Pharma Plant Mission
- Brahmaputra Biodiversity and Biology Boat (B4): Under this program, large boats will be set up in the river with a well-equipped laboratory and cold storage facility to store samples.
4. Atal Jai Anusandhan Biotech Mission
- Atal Jai Anusandhan Biotech Mission has started with the aim of transforming the health, agriculture and energy sectors in the next 5 years.
- It has four components
- GARBH-ini: To promote Maternal and Child Health
- IndCEPI: To develop affordable vaccines and biofortified wheat to promote nutritional security.
- Mission on Anti-Microbial Resistance
- Clean Energy Mission: To promote the invention of innovative technologies to promote Swachh Bharat.
5. BioE3 (Biotechnology for Economy, Environment and Employment) Policy
- It aims to foster high performance biomanufacturing and transforming the existing industrial processes to make them environment friendly and more sustainable.
- The policy focuses on 6 strategic sectors i.e. 1. BIO-BASED Chemicals and Enzymes 2. Functional Foods and Smart Proteins 3. Precision Biotherapeutics 4. Climate Resilient Agriculture 5. Carbon Capture and 6. Futuristic Marine and Space Research.
- The policy also focuses on setting up several biomanufacturing hubs across India.
- Updates (2025): Department of Biotechnology has decided to fund a research project to develop ‘SMART PROTEINS’ in the laboratory under the BioE3 Project to supplement the diet in Indian households
6. Foreign Direct Investment
- 100% FDI is allowed for Greenfield Biotechnology Projects.
- 74% FDI is allowed for Brownfield Biotechnology Projects.
7. Other Steps
- Biotechnology Industry Research Assistance (BIRAC): It is a Public Sector Undertaking of Department of Biotechnology.
- To financially support the Biotech Industry, the government has set up a Venture Capital Fund.
- Patents are essential for biotech companies. Indian Parliament has passed the Patents Act, which allows a 20-year patent term in line with the WTO and TRIPS provisions.
- The government is going to amend the Drugs and Cosmetics Act to make it more compatible with the international standards to attract Biotech companies in this sector.
Challenges faced by Biotechnology Sector
- The Biotechnology Regulatory Authority Bill to create a regulatory body for the Biotech sector is pending in the parliament since 2008.
- Issues with Intellectual Property Regime of India
- Stricter standards of Patents and Intellectual Property in India acts as a hindrance to foreign investment.
- Provision of compulsory licensing in India act as a hindrance as well.
- Jobs offered in the Indian job market are lesser than the workforce supply available.
- Biotech Startups are not able to get early-stage funding.
- India is fast losing to competition created by China and Korea due to regulatory and infrastructure challenges.
- R&D activities are abysmally low in Indian biotechnology. Most of the industry is involved in the manufacture of outsourced products. R&D expenditure is also very low in India
- India = 0.67% of GDP
- Japan and USA = 3% of GDP
- China = 2% of GDP
- Most of the development has happened in the BioPharma sector (drugs & vaccines) only (~55%), while other sub-sectors within Biotechnology have been neglected.
- IPR protection issues: Biotech (especially Biopharma) companies demand TRIPS+ protection & are worried about provisions like Compulsory provisioning.
GEAC (Genetic Engineering Appraisal Committee)
- GEAC works under the Ministry of Environment, Forest and Climate Change and was established under the provisions of GM Rules 1989 of the Environment Protection Act.
- GEAC is responsible for granting approvals to release genetically engineered organisms and products into the environment, including experimental field trials. It can also punish someone found guilty for environmental damage under Environment Protection Act, 1986.
Problem with present Process
- Environment Ministry has a veto over the decisions of GEAC. In UPA Regime, Environment Minister Jairam Ramesh vetoed BT Brinjal even after approval by GEAC.
- Since Agriculture is a State Subject, States can refuse approval even after approval by the Environment Ministry.
- GM crop is an agricultural issue, but the Ministry of Environment is the nodal ministry.
Biotechnology Regulatory Authority of India (BRAI) Bill, 2013
- Biotechnology Regulatory Authority of India (BRAI) will replace GEAC.
- It will consist of chairperson & 5 members who can be eminent scientists or government officials with experience in the field.
- It also established an Appellate Authority.
- The single body will be responsible for any use and clinical trials of Genetically Modified crops, vaccines, drugs etc.
Issues with Bill
- The Bill says that the Tribunal will have jurisdiction over ‘substantial question regarding modern biotechnology’ but fails to explain the term. On the positive side, keeping the term undefined gives flexibility to the judiciary but increases the ambiguity.
- Technical Members of the Tribunal can be eminent scientists or government officials. But the technical expertise of both isn’t the same.
- The Bill doesn’t have the provision of financial liability in case of damage.
Note: BT-Cotton is the only approved GM crop (2002) for commercial cultivation. In 2009, Bt-brinjal was cleared by GEAC for commercial cultivation, but it was put on a 10-year moratorium following public backlash.
Side Topic: Approved GM Crops
- BT-Cotton is the only approved GM crop (2002) for commercial cultivation.
- In 2009, Bt-brinjal was cleared by GEAC for commercial cultivation, but it was put on a 10-year moratorium following a public backlash.
- DMH-11’s (GM Mustard) release into the environment was also placed on hold after the Supreme Court’s pronouncement, although the government had approved its release into the environment.
